Date First Published: October 28, 2014
Date Last Revised: July 3, 2017
Role Of Quality Measurement and Control
In any production system there is a tendency towards variation in quality due to:
- Natural variation due to chance
- Failure to conform to set specifications
The role of quality control is to reduce or eliminate variations that are outside acceptable limits. Thes variations cannot be known unless they are measured and compared with what is expected.
DEFINITION OF QUALITY CONTROL (QC)
A good definition of Quality Control is given by J.M Juran, in his book ‘Quality by Design’, published by Free Press:
“The regulatory process through which we measure the actual performance, compare it with standards and act on the difference”
ISO (ISO 8402) defines it as:
“ The operational techniques and activities (e.g. sampling, testing, documentation of test results etc) that are used to fulfill requirements for quality”.
This is less helpful. Quality control is not meant primarily for fulfilling a requirement.
The standards mentioned in the definition refers primarily to the end product (in the case of manufacturing) or the outcome (in the case of services). For manufactured products, it is possible to select for delivery (ship, sell) only products that are fit for use by rejecting and throwing away defective products. The customer also has the choice to stop using or discard the product that is not useful. For a service product, the implication is quite different. The customer has to experience the service first; but once given, the experience and effect cannot be retracted. Therefore the quality has to be right the first time and every time.
Health care involves both the delivery of services and provision of products (e.g. drugs, food and blood products). While it is necessary that the items given must be of certain quality, the choice of the product and the manner, quantity and timing are decided by care providers and are therefore important aspects of the service. Hence, not only the final outcome is important but also the intermediate outcomes and the way the entire service is delivered.
MEASUREMENT OF QUALITY
To know whether an acceptable quality been achieved, the level of quality need to be known by measuring it. The challenge is to determine what to measure and how to measure it.
An understanding of the determinants of quality based on the Systems Theory of Operations Management discussed in an earlier article is essential.
Measuring Service Quality
The quality of a service is more difficult to measure compared to that of tangible products. To measure quality of services, the following is required:
- Clear definition of quality
- Identification of parameters (quality characteristics) to measure
- Valid measuring tool, valid scales & units of measure
- Set standards (acceptable/tolerance levels or control limits)
Definition of Service Quality
There are three main ways of defining quality i.e.
- Satisfying customer needs
- Fit for use
- Conformance to standards and specifications
These three definitions are not just different ways of looking at quality but in terms of Quality control, each refers to different levels where control need to be put in place. The first relates to the overall effect of the entire service on the customer. The second focuses on functionality, a major characteristic of quality; which in health care is equated with effectiveness. The third stresses on the need to put in place the input, process and environment required to achieve the final outcome.
Dimensions of Quality
Quality Control need to be applied not only to the outcome but also the process, input and environment. The characteristics to measure, the ways of measuring them and the standards need to be defined for each level. Also, the choice of the (key) characteristics to measure and control is dependent on needs, purposes and objectives. These characteristics may (effectiveness, efficiency, safety) not appear to be measurable in a precise manner but must be converted to objective parameters that can be counter or measured.
The use of Quality Control is at two levels:
- Operations level – applied to individual customer or service instance (identifying instances of success or failure)
- Systems design level – applied to the entire group of customers of the service product or all instances (incidence in a population -statistical quality control)
For purposes of improving a service the performance for the entire group (the population), or at least a representative sample of customers receiving the service must be known. However, a population is made up of individuals. Therefore, the outcome achieved for each individual must be measured and documented first. More importantly, a conscientious care provider would set the optimal expected outcome for each individual patient and strive towards achieving it.
Purpose of Quality Control
QUALITY OF THE FINAL OUTCOME
The Need to Measure the Quality of Outcome
In health care delivery, as in any service, the level of quality of the final outcome is most valued by the customer. The end product or outcome need to be measured for the following reasons:
- to know whether the objectives (standards) have been achieved
- to provide comparison with previous and future performance
- to allow comparison with others
- to know the degree of inadequacy or shortcoming
- provide feedback for quality improvement
Often the quality of outcome cannot be measured directly. Instead, surrogate (representative) values are used.
Quality Characteristics of Outcome
The desired outcome of patient care differs from disease to disease and from patient to patient. The characteristics that matter most to patients i.e. their Primary needs are:
- Effectiveness
- Adequacy
- Sustainability
- Safety
The above characteristics become the criteria used for distinguishing an acceptable from an unacceptable outcome.
Measurement & Control of Effectiveness
The patient wants their health problem to be resolved. Their expectation is for their illness to be healed or cured. Yet, this is not possible all the time. In health care, “effectiveness” is defined by the most optimal that can be achieved for a given health problem given the natural history of the illness, the stage at which treatment is sought or given and the patient’s inherent capacity to respond to the illness and treatment. Therefore, effectiveness can vary from cure to obtaining relief of symptoms to experiencing comfort.
“to cure sometimes, to relieve often to comfort always“
attributed to Hippocrates
Ambroise Paré (French Surgeon 1510-1590)
Edward Livingston Trudeau, 19th-century American physician
William Osler,
Standards of Effectiveness
In most instances (based on experience and research), the expected outcome of a given disease is known. To compare results between providers or institutions or for different periods within the same institution, the outcome of a population of patient is a more appropriate standard to use. This is usually a range because of the variation between patients and in the stage, grade and severity of the disease. The width of this range (degree of variation in the outcome) is dependent on the heterogeneity of the group of patients (the population). When patients are more alike (heterogenous) this range is narrow. The measurement of quality for a population of patients is performed through the activity of Medical audit.
However, it must be remembered, that a population is made up of individuals. Therefore the outcome achieved for each individual must be measured and documented first. More importantly, a conscientious care provider would set the optimal expected outcome (the standard) for each individual patient and strive towards achieving it. This standard is based on a set of specifications (often using a scoring system). The success or failure to achieve the expected outcome must be documented. One way is to use the mechanism of incidence reporting. The topic of outcome documentation is discussed further in the article on ‘Monitoring, Review, Assessment and Continuity’.
Measurement and Control of Adequacy
Adequacy refers to the completion of all that is necessary to be provided or performed. The comprehensiveness of the service provided is discussed in the article on “Clinical Care Processes and the Total Patient Care Concept”. To measure adequacy, the components of care are itemized when the Standard Operating Procedure or Care Plan is designed. It is a measure of adherence to plan. At a gross level, control involves making sure that list of tasks is carried out as planned. At a finer level, there is a need to ensure that the content of each item in the service (the intermediate products) is sufficiently provided.
The method of control would involve not only the use of checklists to ensure completeness but also determining the adequacy of content of each process. The latter would require observations of processes as they are performed or an interview of the patient/carers. The topic of ‘SOP and Care Plan’ is discussed in another article.
Measurement & Control of Sustainability
Sustainability is the guarantee that what has been achieved has a certain degree of permanence.
The degree of permanence is dependent on the nature of the illness and whether recurrence can be prevented or otherwise. This does not necessarily mean the termination of the disease or problem. For diseases such as infection or malignancy, permanence (cure) is achieved if all malignant cells or organisms are eliminated. For chronic illnesses, sustainability of the treatment corresponds to maintenance of the optimal health status. Quality Control is used to assess the care provider’s ability to provide services that enable patients to maintain acceptable level of various physiological, psychological or social parameters, avoid triggering factors and follow conducive lifestyle and habits. Therefore, the measure of sustainability can be:
- the frequency of recurrence
- disease free interval
The care provider need to work towards promoting the persistence of wellness and avoidance of the worsening of health (recurrence, development of complications). These actions are termed as ‘continuity of care’ and include:
- regular follow up (monitoring, review & reassessment)
- ensuring compliance
Hence, actual and surrogate (representative) measures of sustainability include:
- The duration of absence of symptoms or effects of the disease (the disease free interval)
- Re-admission rates
- Frequency of occurrence of exacerbation (e.g. in Bronchial Asthma or Psoriasis)
- Degree of compliance (through self-reporting by the patient or using indirect measures)
Measures that demonstrate the adherence to plans e.g. compliance to the follow up appointments, performance of review using key tests (such as HbA1c for diabetes) as scheduled, are examples of indicators of quality of process.
Measurement & Control of Safety of the End Product
Safety is more of an issue relating to the process of care rather than outcome. Yet, there are instances when the final effect of treatment may give rise to increased risks to the health or life of the individual. Before providing a certain treatment method, due consideration need to be made on risks to the patient’s safety. Specific examples are leaving an organ donor with only one kidney, giving Warfarin to a patient after replacing a heart valve and putting a patient on life-long immuno-suppressant drug. In such circumstances the additional care to avert the danger need to be put in place.
QUALITY CONTROL OF PROCESSES
The Need to Measure the Quality of Processes
The work of providing care is achieved mainly through the performance of procedures (jobs, tasks) which are further broken down into processes. How the processes are to be performed and what equipment is to use and the persons eligible to perform the processes are determined when the service delivery system is designed, and the standard operating procedures are developed. In other words, there is a prescribed way of performing processes which needs to be conformed to.
How processes are performed affects all aspects of service delivery operations including:
- Productivity
- Efficiency
- Quality
- Safety
Indeed, efficiency (speed and timeliness) and safety are considered as quality characteristics of a service.
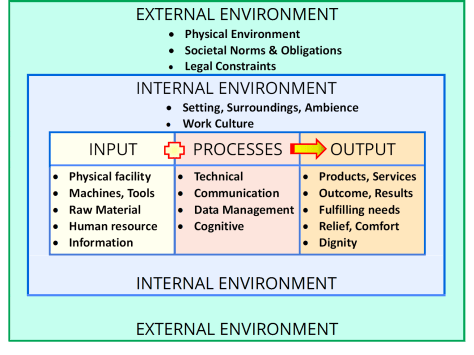
How Processes Transform Input into Output
The conformance of the quality of processes to acceptable standards is determined through two main methods:
- observation of the conduct of the process
- evaluation of the characteristics of the intermediate product
At the individual patient level, the quality of processes and the output of the process (intermediate output/outcome) is measured to ensure the following:
- they are performed according to stipulated (design) specifications so that the desired outcome is achieved (the standard operating procedure)
- the well-being and safety of the patient while undergoing the process
- the whole experience (physical, psychological and social) is as pleasant, dignified and acceptable as possible
Thus for a patient, each process need to be under constant scrutiny to ensure that each subsequent step proceeds only if the procedure thus far is going according to plan. Like assembly lines the entire process of a service can be stopped and resumed only after adjustments are made to faulty or inadequate input (equipment, instruments, materials, human resource) or the environment made more conducive. In other words, a method of treatment is done after all requisites has be put in place or is available. For example, to give inotopic drugs to a patient in cardiogenic shock, requires that the drug is given by micro-infusion, the heart rate, blood pressure is continuously monitored and there is a trained care provider constantly monitoring the parameters. Such a requirement is available in an ICU setting. If the patient is to be treated in an ordinary ward, then certain actions must be taken to meet the requirement.
In services, once given, the final outcome (both the benefits and harm encountered) cannot be undone, reworked or recalled. However there is, in most instances, a window of opportunity to somehow seek a different path, redress the shortcomings, repair some of the damage and recover some of the losses. Control mechanisms of processes or intermediate products such as Progress Review and Re-evaluation can help improve the final outcome. If the treatment as planned proved to be ineffective or inappropriate, the SOP or Care Plan may be redesigned. If a defect is discovered, damage control through efforts such as amelioration of side effects and tertiary preventive measures (e.g. rehabilitation) can be done. If there is error of omission, it may not be too late to perform the necessary task. Quality control of the care of an individual patient is more useful if it is prospective rather than retrospective.
However, knowing how a care provider or a service (team, unit, hospital) fare when caring for a group of patients (a population) is also important for the following purposes:
- to enable comparisons to be made
- with others or a benchmark
- with the performance at an earlier period
- to provide feedback for improvement of service delivery systems, processes and product design
Measuring the level of quality of processes in a population is called Statistical Process Control. In healthcare. the main method is Medical Audit.
Quality Characteristics of Processes
A process does not stand on its own. Instead, a set of various types of processes make up a procedure. Procedures and processes can be divided based on their purpose and the method used into:
- Technical Process
- Communication
- Cognitive Process
- Data Management Process
- Administrative Process
A procedure is made up of a combination of the above process types. Measurement of quality characteristics may be performed at the procedure or the process level. Each of the types has a different set of quality characteristics. Also quality of process is perceived differently from the point of view of the customer as opposed to the provider.
Measurement & Control of Technical Processes
A technical process is a manual act performed with or without the aid of tools or instruments. It could also be completely mechanized and automated.
Quality characteristics of technical processes valued by customers include:
- Precision
- Efficiency
In health care, patients would like to experience processes that gets the intended output (get it right) with minimal efforts; preferably ‘first time every time’. They would appreciate if it is performed on time and taking the shortest time.
For the care provider, control measures should assist him/her to achieve the intended outcome. Control here means ensuring conformance to the process design i.e. knowing that the procedure or process is performed the way it should be (e.g. based on the SOP), preventing it from going wrong and correcting any mistakes. Achieving control depends on two factors:
- The desire and ability of the person performing to do it right
- Supervision
Therefore, the quality of processes performed is dependent on the knowledge, skills and attitude of the operator (care provider or technician). In healthcare where care providers are constantly learning new tasks. Therefore, supervision must be in place during training such that the supervisor can assess and influence the performance of the trainee.
Measuring conformance requires a clear description of procedures and processes. This is achieved by documenting them as Standard Operating Procedures.
From the perspective of operations management, the manager (or anyone responsible for quality management and system design) would want to know the frequency of conformance to the process for all instances of service delivery. The quality control data would allow for the calculation of frequencies and rates (percentages, proportions). He/she would then be able to gauge:
- the capability of workers to perform processes
- which particular process(es) is poorly performed
- which procedure/process fails to produce the intended outcome
The data made available, would provide feedback to the manager (quality manager, supervisor) and help him/her devise means of improving conformance (statistical quality control) usually through training, supervision or motivation.
Through Statistical process control the system designer (technical expert or specialist) evaluates which procedure/process fails to produce the intended outcome. Following that a more detailed study is carried out by:
- observing the process as it is being performed,
- studying the outcome of the process (the intermediate outcome)
- obtaining feedback from the worker who performs the process.
In this way the inherent characteristic of the process (as it is practiced) is known and the information is used to improve its design. The following characteristics should be measured:
- Effectiveness
- Reproducibility
- Ease of performance
- Efficiency (Optimal use of resources)
Measurement & Control of Communication as a Process
Communication between care providers with each other and with patients is an important aspect of service delivery. It impinges on effectiveness and safety of the final outcome.
Communications between care providers ensure information is shared, intentions are made clear, and processes are streamlined.
Good communication puts patients at ease, promote compliance and enhance the whole service experience. They would appreciate communication that is effective, friendly, respectful, adequate and clear. Communication requires the right skill, knowledge and attitude.
Control of the quality communication during the provision of care of an individual patient is difficult. A good method to gauge clarity and adequacy of patient education, briefing and instruction, is to seek a response from the patient whether he/she understands them. In some instances, he/she may be asked to repeat the instruction given.
The measurement of quality of communication provided to a patient population is gauged by through Customer Satisfaction Survey including it as one of the criteria. This is more useful if the survey is conducted for a specific service product.
Measurement & Control of Productivity, Efficiency and Quality
Operational Systems in Service Delivery
It is also important to ascertain that equipment used is in proper functioning order and staff with appropriate knowledge and skills are assigned to perform the processes. The atmosphere / environment also need to be controlled. Therefore, quality control is more beneficial if it is prospective (preventive) rather than retrospective (reactive) in approach.



great great article ,thanks
LikeLike
Nice share ! Thanks for the post in quality control
https://amrepinspect.com/quality-control-inspection-services/
LikeLike